
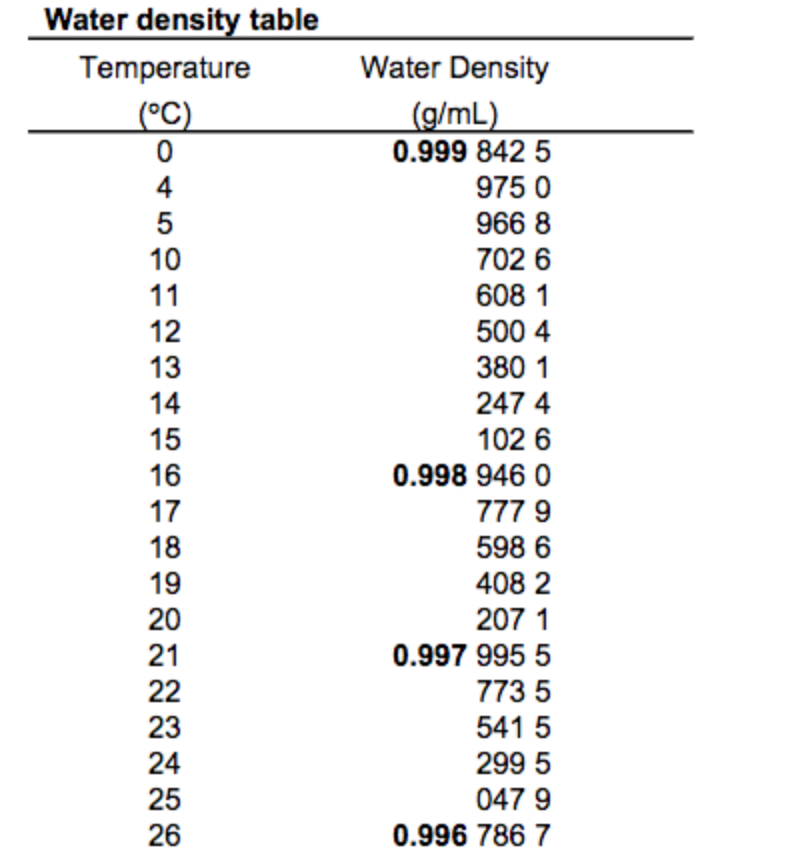
This unusual negative thermal expansion is related to the strong intermolecular forces or interactions and it is observed in the form of molten silica.As it gets cooled further, the water tends to expand and becomes less dense.Approximately at about 4☌, pure water reaches its maximum density. When water is cooled from room temperature, it tends to become increasingly dense.
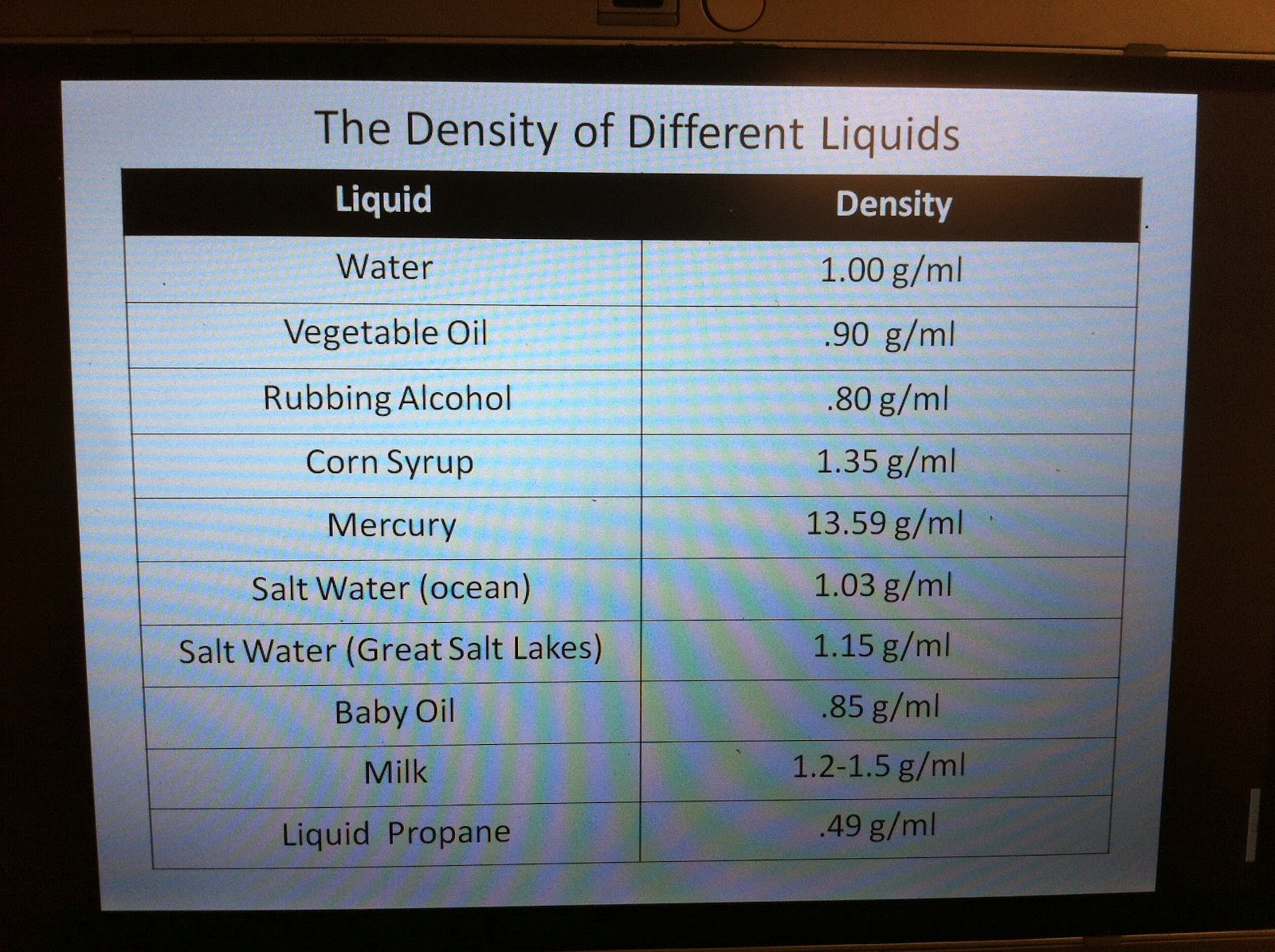
When the temperature goes down, the seawater becomes dense. With an increase in temperature, the water gets warmer and molecules open up.The density of water is approximately 1 gram/centimeter (1 g/cm 3). It is temperature-dependent, but this relation is -linear and unimodal in nature instead of monotonic.It is denser than pure water as it contains many chemicals packed within it. Seawater density also varies with depth and temperature. Seawater may be an example of saline water. For example, in the case of saline water, an increase in salinity makes the water dense. In the case of impure water, water density increases with reference to impurity.Due to this structure, ice is less dense as compared to liquid water. It allows the water molecules to make more hydrogen bonds preventing the molecules from returning closer to each otherĪ hexagonal structure forms and leaves some space between the molecules which leads to a decrease in density. In pure water, lowering the temperature reduces thermal motion.Three factors that affect the density of water are as follows: So, the density of water at 4 0C is the maximum and the specific volume is the minimum.ĭifference between gravitation and gravity At 4 oCĪt a temperature of 4 degrees, the thermal expansion and the hydrogen bond formation tend to balance each other in order to give a stationary behavior for the density. This prevents the water molecules from coming closer and therefore the density of water decreases. Due to the lack of thermal motion, more and more hydrogen bonds are formed.

In this range of temperature, the water molecules come closer and they are able to make stable Hydrogen bonds. The molecules come closer and therefore the density increases. When water is cooled from a high temperature, the thermal energy of the molecules of water reduces. When the temperature decreases, water converts into solid, and when the temperature increases, water converts into gas. Water is abundantly available in the world and is found in three forms ( solid, liquid, and gas). The density of a substance is usually measured in grams per cubic centimeter. Density of Water Formulaĭensity is calculated by the formula d = M/V, where d is density, M is mass, and V is volume. At normal temperature, water has a density of 0.99823 g/ml. Water does not have any absolute density as the density of water varies with its temperature.
Density describes how much space an object takes up in relation to its mass.
